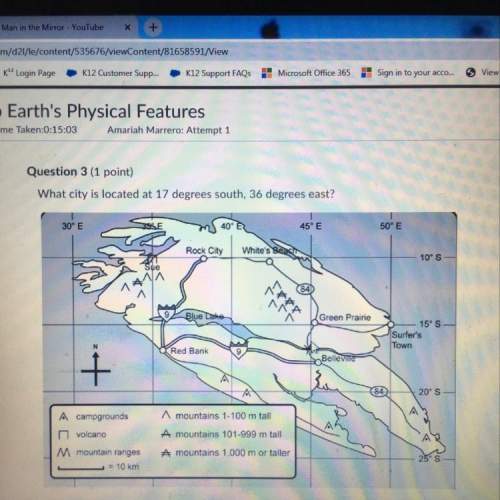
Consider the chemical equations shown here. Upper N Upper O (g) + Upper O Subscript 3 Baseline (g) right arrow Upper N Upper O Subscript 2 Baseline (g) + Upper O Subscript 2 Baseline (g) delta Upper H Subscript 1 Baseline = negative 198.9 kilojoules. StartFraction 3 Over 2 EndFraction Upper O Subscript 2 Baseline (g) right arrow Upper O Subscript 3 Baseline (g) delta Upper H Subscript 2 Baseline = 142.3 kilojoules. Upper O (g) right arrow one half Upper O Subscript 2 Baseline (g) delta Upper H Subscript 3 Baseline = negative 247.5 kilojoules. What is Delta. Hrxn for the reaction shown below? Upper n upper O (g) plus upper O (g) right arrow upper N upper O subscript 2 (g).

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 23.06.2019 01:30
Will a solution form when the solvent and solute are both nonpolar? a. not likely b. never c. most likely
Answers: 1

Chemistry, 23.06.2019 03:50
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer
Answers: 1

You know the right answer?
Consider the chemical equations shown here. Upper N Upper O (g) + Upper O Subscript 3 Baseline (g) r...
Questions

History, 18.10.2021 03:40

Mathematics, 18.10.2021 03:40


Mathematics, 18.10.2021 03:40



Mathematics, 18.10.2021 03:40

Mathematics, 18.10.2021 03:40


Mathematics, 18.10.2021 03:40


Spanish, 18.10.2021 03:40

Health, 18.10.2021 03:40



History, 18.10.2021 03:50


English, 18.10.2021 03:50