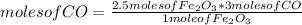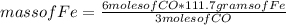Consider the process used to produce iron metal from its ore.
Fe2O3(s) + 3CO(g) --> 2Fe(s) + 3CO2 (g)
How many grams of iron can be produced from 2.5 moles of Fe2O3 and 6.0 moles of CO? Hint: limiting reactant problem
O A. 140 g
B. 335 g
C. 55.858
D. 223 g Fe
E. 279 g Fe

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3



Chemistry, 23.06.2019 02:00
What is the difference between a substance "getting wet" and "being dissolved" in a liquid at the particulate level?
Answers: 3
You know the right answer?
Consider the process used to produce iron metal from its ore.
Fe2O3(s) + 3CO(g) --> 2Fe(s) + 3CO...
Questions


Mathematics, 11.11.2021 02:50


Physics, 11.11.2021 02:50

English, 11.11.2021 02:50

Advanced Placement (AP), 11.11.2021 02:50

Mathematics, 11.11.2021 02:50



SAT, 11.11.2021 02:50

Mathematics, 11.11.2021 02:50


Business, 11.11.2021 02:50

Chemistry, 11.11.2021 02:50





Biology, 11.11.2021 02:50

Mathematics, 11.11.2021 02:50