
Chemistry, 06.12.2020 19:00 alanflores40
The pH of a 0.15 M butylamine, C&H3NH2 solution is 12.0 at 25°C. Calculate the dissociation constant of the base.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Balance this equation co2(g) + h2o (g) show that the balanced equation obeys the law if conversation of mass
Answers: 1

Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 10:50
Determine the empirical formula for succinic acid that is composed of 40.60% carbon, 5.18% hydrogen, and 54.22% oxygen.
Answers: 1

Chemistry, 22.06.2019 13:00
Asubstance is a good conductor of electricity which of the following best explains a probable position of the substance in a periodic table
Answers: 3
You know the right answer?
The pH of a 0.15 M butylamine, C&H3NH2 solution is 12.0 at 25°C. Calculate the dissociation
con...
Questions

Chemistry, 08.10.2019 16:40

Computers and Technology, 08.10.2019 16:40

Mathematics, 08.10.2019 16:40


History, 08.10.2019 16:40

Business, 08.10.2019 16:40


Physics, 08.10.2019 16:40

Mathematics, 08.10.2019 16:40

Social Studies, 08.10.2019 16:40


English, 08.10.2019 16:40






Mathematics, 08.10.2019 16:40

History, 08.10.2019 16:40

Physics, 08.10.2019 16:40

![\rm Kb=\dfrac{[L][OH^-]}{[LOH]}](/tpl/images/0954/6725/84d1f.png)
![\tt [OH^-]=\sqrt{Kb.M}](/tpl/images/0954/6725/ab388.png)
![\tt [OH^-]=10^{-pOH}\\\\(OH^-]=10^{-2}](/tpl/images/0954/6725/b2b50.png)
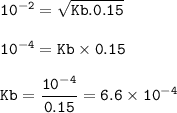
![\tt Kb=\dfrac{x^2}{0.15-x}\rightarrow x=[OH^-]\\\\Kb=\dfrac{10^{-4}}{0.15-10^{-2}}=7.14\times 10^{-4}](/tpl/images/0954/6725/69346.png)


