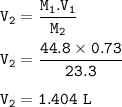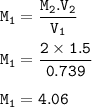Chemistry, 05.12.2020 14:00 najeezubair0666
Part 1: What is the final volume in milliliters when 0.730 L of a 44.8 % (m/v) solution is diluted to 23.3 % (m/v)? part 2:A 739 mL NaCl solution is diluted to a volume of 1.50 L and a concentration of 2.00 M . What was the initial concentration?

Answers: 1
Another question on Chemistry


Chemistry, 23.06.2019 00:10
In as 1°, 2°, 3°, or 4°. be to . : °b: °c: °d: ° : °b: °c: °d: ° : °b: °c: °d: °e: °f: °g: °h: ° : °b: °c: °d: °e: °f: °g: °h: °i: °
Answers: 3

Chemistry, 23.06.2019 03:30
The molar mass of iron(fe) is 55.8 g/mol. what is the mass in grams of 2.25 moles of iron?
Answers: 1

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1
You know the right answer?
Part 1: What is the final volume in milliliters when 0.730 L of a 44.8 % (m/v) solution is diluted t...
Questions


Chemistry, 17.11.2020 23:40

Social Studies, 17.11.2020 23:40

Mathematics, 17.11.2020 23:40



Mathematics, 17.11.2020 23:40

Mathematics, 17.11.2020 23:40

Mathematics, 17.11.2020 23:40


Business, 17.11.2020 23:40

Arts, 17.11.2020 23:40


Mathematics, 17.11.2020 23:40

Arts, 17.11.2020 23:40

Mathematics, 17.11.2020 23:40