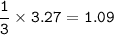Chemistry, 05.12.2020 03:50 nell1234565
How many moles of nitrogen gas would be produced if 3.27 moles of copper(II) oxide were reacted with excess ammonia in the following chemical reaction? 2 NH3(g) + 3 CuO (s) – 3 Cu(s) + N2(g) + 3 H2O(g)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 12:00
Most materials are not magnetic because their magnetism has worn off. their magnetic domains are arranged randomly. they lack magnetic fields. earth’s heat has destroyed their magnetism.
Answers: 1

Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1
You know the right answer?
How many moles of nitrogen gas would be produced if 3.27 moles of copper(II) oxide were reacted with...
Questions


Biology, 20.09.2020 15:01

Biology, 20.09.2020 15:01


Mathematics, 20.09.2020 15:01





Business, 20.09.2020 15:01

Mathematics, 20.09.2020 15:01

Social Studies, 20.09.2020 15:01

English, 20.09.2020 15:01


Mathematics, 20.09.2020 15:01

History, 20.09.2020 15:01

Chemistry, 20.09.2020 15:01

Mathematics, 20.09.2020 15:01

Mathematics, 20.09.2020 15:01

Physics, 20.09.2020 15:01