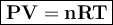Please help fast
A person breathes in 6.0 L of pure oxygen at 298 K and
1,000 kPa to fill the...

Please help fast
A person breathes in 6.0 L of pure oxygen at 298 K and
1,000 kPa to fill their lungs
How many moles of oxygen did they take in?
Use the ideal gas law: PV = nRT where R = 8.31 L – kPa /mol – K
A) 0.05 mole
B) 0.41 mole
C) 2.42 moles
D) 20.0 moles

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:00
2h2s + 3o2 2so2 + 2h2o which option gives the correct mole ratios? h2s: so2 = 2: 2 and o2: h2o = 3: 2 h2s: so2 = 2: 3 and o2: h2o = 3: 2 h2s: so2 = 4: 4 and o2: h2o = 5: 4 h2s: so2 = 4: 6 and o2: h2o = 4: 4
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 17:30
Aroller coaster is traveling at 13 mi./s when you purchase a hill that is 400 m long and down the hill exonerate at 4.0 m/s squared what is the final velocity of the posterior found your answer to the nearest number
Answers: 1

You know the right answer?
Questions

Mathematics, 16.12.2020 20:40

Mathematics, 16.12.2020 20:40

Mathematics, 16.12.2020 20:40

Biology, 16.12.2020 20:40



Biology, 16.12.2020 20:40

Mathematics, 16.12.2020 20:40



Chemistry, 16.12.2020 20:40


History, 16.12.2020 20:40


Mathematics, 16.12.2020 20:40


Mathematics, 16.12.2020 20:40

Physics, 16.12.2020 20:40

Geography, 16.12.2020 20:40