How many oxygen atoms are in the product of this balanced chemical equation
...

Chemistry, 05.12.2020 01:00 madison6592
How many oxygen atoms are in the product of this balanced chemical equation
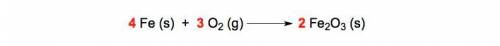

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 23.06.2019 01:30
Select the correct answer from each drop-down menu. to make a table of the elements, dmitri mendeleev sorted the elements according to their . he then split the list of elements into several columns so that elements beside each other had similar .
Answers: 2

Chemistry, 23.06.2019 08:00
The goal of this experiment was to answer the question "what is the effect of a gas' temperature on its volume? " you formulated the hypothesis below. hypothesis: if a fixed amount of gas is heated, then the volume will increase because the heat will cause the molecules of gas to move faster and further apart. to test this hypothesis, you changed the of the gas between 0 and 100°c (273 and 373 k) and calculated the resulting of the gas.
Answers: 2
You know the right answer?
Questions


Mathematics, 14.02.2021 03:50


Mathematics, 14.02.2021 03:50

Mathematics, 14.02.2021 03:50

Mathematics, 14.02.2021 03:50


Mathematics, 14.02.2021 03:50

Chemistry, 14.02.2021 03:50

Mathematics, 14.02.2021 03:50

Mathematics, 14.02.2021 03:50




Computers and Technology, 14.02.2021 03:50

Mathematics, 14.02.2021 03:50



Computers and Technology, 14.02.2021 03:50



