
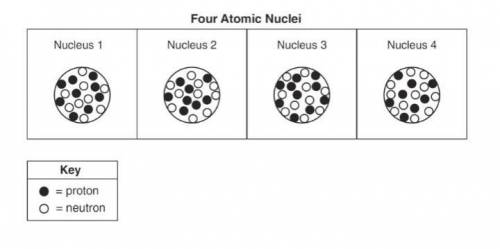
Base your answers to questions 22, 23, and 24 on the information below and on your knowledge of chemistry. The diagrams represent four different atomic nuclei.
22. Identify the element that has atomic nuclei represented by nucleus 1.
23. Determine the mass number of the nuclide represented by nucleus 2.
24. Explain why nucleus 2 and nucleus 4 represent the nuclei of two different isotopes of the same element.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 08:30
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2
You know the right answer?
Base your answers to questions 22, 23, and 24 on the information below and on your knowledge of chem...
Questions

Arts, 19.04.2021 20:30

Mathematics, 19.04.2021 20:30



Chemistry, 19.04.2021 20:30

History, 19.04.2021 20:30


English, 19.04.2021 20:30

Mathematics, 19.04.2021 20:30

Social Studies, 19.04.2021 20:30


Mathematics, 19.04.2021 20:30



English, 19.04.2021 20:30

Mathematics, 19.04.2021 20:30

Mathematics, 19.04.2021 20:30

History, 19.04.2021 20:30

Chemistry, 19.04.2021 20:30

Mathematics, 19.04.2021 20:30



