
Chemistry, 03.12.2020 20:10 harris435942
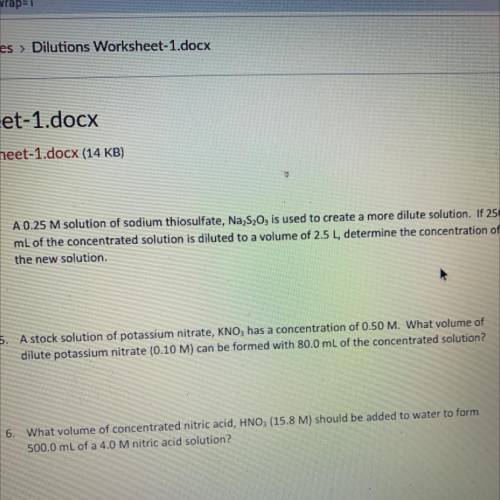
A stock solution of potassium nitrate, KNO3 has a concentration of 0.50 M. What volume of dilute potassium nitrate (0.10 M) can be formed with 80.0 mL of the concentrated solution?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 01:20
Match the acid base pairs by arranging the acid name with the conjugate base formula. hydrogen carbonate hydrogen phosphate carbonic acid read water sulfuric acid phosphoric acid a. co32- b. hso4- c. hco3- d. po43- e. h2po4- f. oh-
Answers: 1


Chemistry, 22.06.2019 19:30
Awoman's basketball has a circumference between 28.5 and 29.0 inches and a maximum weight of 20 ounces (two significant figures). what are these specifications in units of centimeters and grams?
Answers: 2
You know the right answer?
A stock solution of potassium nitrate, KNO3 has a concentration of 0.50 M. What volume of
dilute po...
Questions


History, 26.09.2019 12:00



Biology, 26.09.2019 12:00


Health, 26.09.2019 12:00

English, 26.09.2019 12:00

Physics, 26.09.2019 12:00

Physics, 26.09.2019 12:00


Mathematics, 26.09.2019 12:00


English, 26.09.2019 12:00



Mathematics, 26.09.2019 12:00

Physics, 26.09.2019 12:00




