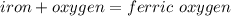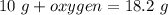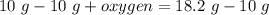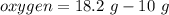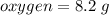Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 23.06.2019 00:30
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1

Chemistry, 23.06.2019 03:50
What is the equation fort the alkaline zinc/manganese dioxide cell. a) anode b)cathode c)overall equations.
Answers: 2
You know the right answer?
A 10 gram sample of iron reacts with oxygen to form 18.2 grams of ferric oxide. How many grams of ox...
Questions



Mathematics, 15.05.2021 04:50

Mathematics, 15.05.2021 04:50


Chemistry, 15.05.2021 04:50

English, 15.05.2021 04:50

Chemistry, 15.05.2021 04:50


Mathematics, 15.05.2021 04:50


Mathematics, 15.05.2021 04:50



Mathematics, 15.05.2021 04:50


Mathematics, 15.05.2021 04:50

Geography, 15.05.2021 04:50

English, 15.05.2021 04:50

English, 15.05.2021 04:50