Chemistry, 02.12.2020 17:10 BrianKeokot7700
a) Calculate the standard reaction entropy for the combustion of methane to carbon dioxide and liquid water at 298 K. b) Calculate the standard reaction entropy for the hydrolysis of liquid water to form oxygen and hydrogen gas. c) Do your results in (a) and (b) agree with the general rule that reaction entropies are positive if there is a net formation of gas in a reaction, and negative if the is a net reduction of gas?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
According to periodic trend, which of the following most likely has the highest ionization energy? kr be ni sc
Answers: 3

Chemistry, 22.06.2019 14:30
An object resting on a table weighs 100 n. with what force is the object pushing on the table? with what force is the table pushing on the object? explain how you got your answer.
Answers: 3

Chemistry, 22.06.2019 23:30
Aweight lifter raises a 1600 n barbell to a height of 2.0 meters. how much work was done? w = fd a) 30 joules b) 3000 joules c) 320 joules d) 3200 joules
Answers: 2

Chemistry, 23.06.2019 01:00
If a sample of radioactive isotopes takes 600 minutes to decay from 400 grams to 50 grams, what is the half-life of the isotope?
Answers: 1
You know the right answer?
a) Calculate the standard reaction entropy for the combustion of methane to carbon dioxide and liqui...
Questions


Mathematics, 27.07.2019 13:00



Mathematics, 27.07.2019 13:00


Mathematics, 27.07.2019 13:00



Chemistry, 27.07.2019 13:00

Mathematics, 27.07.2019 13:00

English, 27.07.2019 13:00




Health, 27.07.2019 13:00




Mathematics, 27.07.2019 13:00

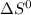 = - 242.2J/K.mol
= - 242.2J/K.mol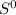 .
. 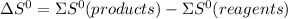
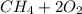 ⇒
⇒ 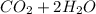
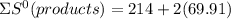 = 353.8
= 353.8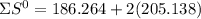 = 596.54
= 596.54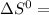 353.8 - 596.54
353.8 - 596.54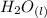 ⇒
⇒ 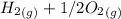
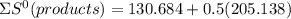 = 233.253
= 233.253