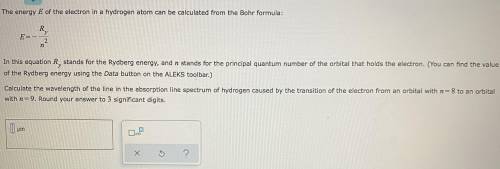
Chemistry, 02.12.2020 17:00 AreYouMyNewDad
g In this equation stands for the Rydberg energy, and stands for the principal quantum number of the orbital that holds the electron. (You can find the value of the Rydberg energy using the Data button on the ALEKS toolbar.) Calculate the wavelength of the line in the emission line spectrum of hydrogen caused by the transition of the electron from an orbital with to an orbital with . Round your answer to significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:50
What are the 4 phases of matter in order of increasing engery content?
Answers: 2

Chemistry, 22.06.2019 06:00
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1
You know the right answer?
g In this equation stands for the Rydberg energy, and stands for the principal quantum number of the...
Questions

English, 04.08.2019 18:30

Chemistry, 04.08.2019 18:30


Mathematics, 04.08.2019 18:30

Computers and Technology, 04.08.2019 18:30

Business, 04.08.2019 18:30



Biology, 04.08.2019 18:30




Spanish, 04.08.2019 18:30


Biology, 04.08.2019 18:30



Biology, 04.08.2019 18:30

Health, 04.08.2019 18:30

Social Studies, 04.08.2019 18:30