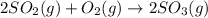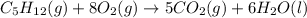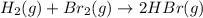Chemistry, 01.12.2020 03:00 AlaskaAirlines
At constant pressure for which of the reactions shown below should Delta H be greater than Delta E°?
1.2 SO2(g) + O2(g) - 2 SO3(g)
II. C5H12(9) + 8 O2(g) - 5 CO2(g) + 6 H20(1)
III. H2(g) + Br2(g) → 2 HBr(9),
IV. N204(9) - 2 NO2(g)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

You know the right answer?
At constant pressure for which of the reactions shown below should Delta H be greater than Delta E°?...
Questions

Mathematics, 13.11.2020 01:10

Mathematics, 13.11.2020 01:10


Mathematics, 13.11.2020 01:10



Mathematics, 13.11.2020 01:10



Mathematics, 13.11.2020 01:10

Mathematics, 13.11.2020 01:10


Mathematics, 13.11.2020 01:10

Mathematics, 13.11.2020 01:10

Mathematics, 13.11.2020 01:10

Mathematics, 13.11.2020 01:10

Mathematics, 13.11.2020 01:10

Mathematics, 13.11.2020 01:10


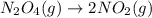 has higher value of
has higher value of 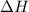 than
than 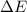
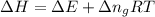
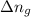 = change in the gaseous moles of the reaction = Moles of product - Moles of reactant
= change in the gaseous moles of the reaction = Moles of product - Moles of reactant