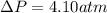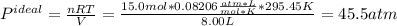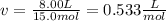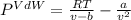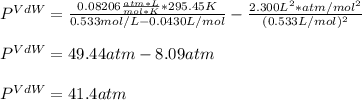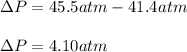Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 18:30
What volume of a 0.0606 m solution of strontium bromide is needed to obtain 0.340 mol of the compound? question 42 options: a)5.61 l b) 3.4 l c) 600 ml d) 1 l e) 178 ml
Answers: 1

Chemistry, 22.06.2019 19:30
Astring vibrates with a frequency of 10 hz. why can't a person hear the sound waves produced by the vibrating string, no matter how large the amplitude of the waves? out! this is homework and due tomorrow! you so much!
Answers: 2
You know the right answer?
15.0 moles of gas are in a 8.00 L tank at 22.3 ∘C∘C . Calculate the difference in pressure between m...
Questions

History, 15.01.2021 03:20


History, 15.01.2021 03:20

Mathematics, 15.01.2021 03:20


Geography, 15.01.2021 03:20

History, 15.01.2021 03:20

Mathematics, 15.01.2021 03:20



English, 15.01.2021 03:20

Mathematics, 15.01.2021 03:20


History, 15.01.2021 03:20

Mathematics, 15.01.2021 03:20


Engineering, 15.01.2021 03:20

English, 15.01.2021 03:20


English, 15.01.2021 03:20