

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:40
Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with solution. calculate the volume of solution the student will need to add to reach the final equivalence point. round your answer to significant digits.
Answers: 3

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2
You know the right answer?
In the reaction, 2HgO(s)⟶2Hg(s) O2(g). How many liters of oxygen, O2 measured at STP, would be produ...
Questions

History, 01.09.2021 15:50







Mathematics, 01.09.2021 15:50


Mathematics, 01.09.2021 15:50


Computers and Technology, 01.09.2021 15:50


Mathematics, 01.09.2021 15:50

Mathematics, 01.09.2021 15:50

English, 01.09.2021 15:50


Health, 01.09.2021 15:50

Mathematics, 01.09.2021 15:50

Mathematics, 01.09.2021 15:50

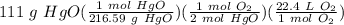 = 5.73988 L O₂
= 5.73988 L O₂

