
Chemistry, 25.11.2020 14:00 maskythegamer
13. How many moles of sodium hydroxide are needed to neutralize 50grams of sulfuric acid? 2NaOH H2SO4 → Na2SO4 + 2H2O

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Which of the following statements concerning the influence of culture on ethnic identity formation is accurate? a. one will reject ethnic identity if cultural stereotypes are encountered. b. if one’s ethnic city is different from the dominant cultural group, then one’s ethnic identity you will become weekend. c. if an the ethnic group is excepted by dominant culture, then ethnic identity formation can be a difficult process. d. similarity to the dominant culture can determine how easy it is for one to except ethnic differences.
Answers: 2

Chemistry, 22.06.2019 01:00
The diagram shows the positions of the sun, moon and earth during spring tides, when the high tides are at their highest and low tides at their lowest. what is it about these positions that causes these high and low tides?
Answers: 3

Chemistry, 22.06.2019 10:00
What is the atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons? a) 6 b) 8 c) + 1 d) 12 e) 18
Answers: 1

Chemistry, 23.06.2019 01:30
Which of the following statements is true about energy quantization at the atomic level? electrons in the outermost orbits are the most stable. electrons in all the orbits around the nucleus have the same amount of energy. electrons in the orbit closest to the nucleus have the least amount of energy. electrons absorb or release the same amount of energy independent of the energy levels.
Answers: 1
You know the right answer?
13. How many moles of sodium hydroxide are needed to neutralize 50grams of sulfuric acid?
2NaOH H2S...
Questions

History, 20.12.2019 18:31

Mathematics, 20.12.2019 18:31

Mathematics, 20.12.2019 18:31



Mathematics, 20.12.2019 18:31

Mathematics, 20.12.2019 18:31



Computers and Technology, 20.12.2019 18:31


English, 20.12.2019 18:31








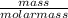
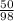 = 0.51mole
= 0.51mole 

