Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
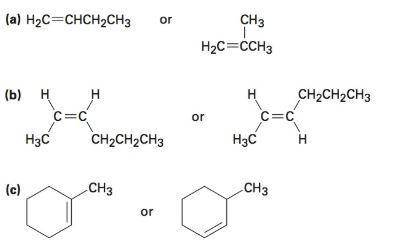
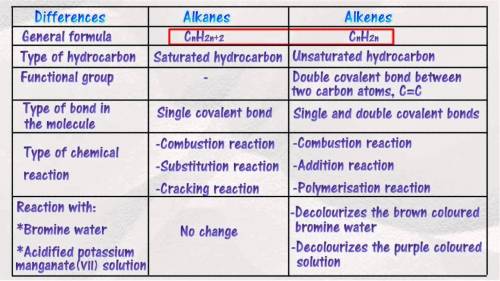
Need images of structures of all alkanes and alkenes...
Questions

History, 20.09.2020 18:01


History, 20.09.2020 18:01


Social Studies, 20.09.2020 18:01

Biology, 20.09.2020 18:01




Mathematics, 20.09.2020 18:01




History, 20.09.2020 18:01

Mathematics, 20.09.2020 18:01



Business, 20.09.2020 18:01

English, 20.09.2020 18:01

History, 20.09.2020 18:01