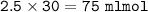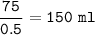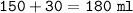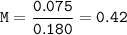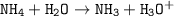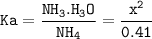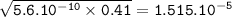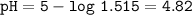Chemistry, 23.11.2020 08:50 raynamg2718
Just enough 0.500 M HCl is added to 30.0 mL of 2.5 M NH3 to reach the equivalence point. The Kb of NH3 = 1.8 X 10-5
Write the balanced equation for this reaction.
What volume of 0.500 M HCl solution was added?
What is the molarity of the salt produced from the neutralization reaction?
What is the pH of the solution at the equivalence point?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:10
What can be added to the examples section of each circle? endothermic: ice melting into water, and a heat pack becoming warm exothermic: a glow stick glowing, and fireworks exploding endothermic: ice melting into water, and an instant ice pack turning cold exothermic: fireworks exploding, and gasoline burning endothermic: a glow stick glowing, and a heat pack becoming warm exothermic: an instant ice pack turning cold, and ice melting into water endothermic: gasoline burning, and an instant ice pack turning cold exothermic: ice melting into water, and an instant ice pack turning cold
Answers: 1

Chemistry, 21.06.2019 22:30
Asample of neon occupies a volume of 375 ml at stp. what will be the volume of neon if the pressure is reduced to 90.0 kpa? a. 422 ml b. 422 l c. 333 ml d. 333 l
Answers: 2

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

You know the right answer?
Just enough 0.500 M HCl is added to 30.0 mL of 2.5 M NH3 to reach the equivalence point. The Kb of N...
Questions




Mathematics, 05.11.2020 19:00



Physics, 05.11.2020 19:00

Social Studies, 05.11.2020 19:00



Mathematics, 05.11.2020 19:00

English, 05.11.2020 19:00


Mathematics, 05.11.2020 19:00



Geography, 05.11.2020 19:00


Mathematics, 05.11.2020 19:00

History, 05.11.2020 19:00