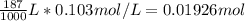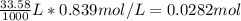Chemistry, 23.11.2020 08:10 eddrekas8564
Consider the following reaction. MgCl2(aq)+2NaOH(aq)⟶Mg(OH)2(s)+2NaC l(aq) A 187.0 mL solution of 0.103 M MgCl2 reacts with a 33.58 mL solution of 0.839 M NaOH to produce Mg(OH)2 and NaCl. Identify the limiting reactant.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Which best describes why nh4+ can form an ionic bond with ci-?
Answers: 1

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2

Chemistry, 22.06.2019 14:00
What term describes technology that operates on an atomic level
Answers: 2

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1
You know the right answer?
Consider the following reaction. MgCl2(aq)+2NaOH(aq)⟶Mg(OH)2(s)+2NaC l(aq) A 187.0 mL solution of 0....
Questions


Arts, 02.11.2020 14:20

English, 02.11.2020 14:20

English, 02.11.2020 14:30

History, 02.11.2020 14:30

Mathematics, 02.11.2020 14:30


Mathematics, 02.11.2020 14:30

Mathematics, 02.11.2020 14:30



Chemistry, 02.11.2020 14:30


Mathematics, 02.11.2020 14:30

Social Studies, 02.11.2020 14:30


Physics, 02.11.2020 14:30


English, 02.11.2020 14:30

Mathematics, 02.11.2020 14:30