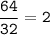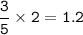Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 02:00
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1

Chemistry, 22.06.2019 04:30
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

You know the right answer?
Given the unbalanced combustion equation: C3H8 + O2 → CO2 + H2O, how many moles of carbon dioxide ar...
Questions



Mathematics, 21.09.2020 19:01


Mathematics, 21.09.2020 19:01

Biology, 21.09.2020 19:01

History, 21.09.2020 19:01

English, 21.09.2020 19:01

Chemistry, 21.09.2020 19:01

History, 21.09.2020 19:01






Mathematics, 21.09.2020 20:01