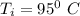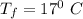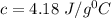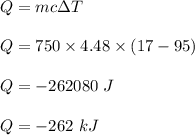Chemistry, 19.11.2020 23:10 ashleymer384
If a 750.0 gram sample of water is cooled for 95.0 oC to 17 oC, how much heat is released? CP = 4.18 J/g oC

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3


Chemistry, 22.06.2019 22:30
What methods could you use to solubilize calcium carbonate
Answers: 1

You know the right answer?
If a 750.0 gram sample of water is cooled for 95.0 oC to 17 oC, how much heat is released? CP = 4.18...
Questions


Mathematics, 09.02.2021 23:10


Chemistry, 09.02.2021 23:10





Mathematics, 09.02.2021 23:10


Mathematics, 09.02.2021 23:10


Mathematics, 09.02.2021 23:10



Mathematics, 09.02.2021 23:10

Mathematics, 09.02.2021 23:10