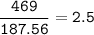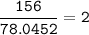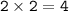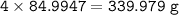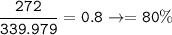Aqueous copper(II) nitrate reacts with aqueous sodium sulfide to produce aqueous sodium nitrate and copper(II) sulfide as a precipitate. In this reaction 469 grams of copper(II) nitrate were combined with 156 grams of sodium sulfide to produce 272 grams of sodium nitrate.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1

Chemistry, 22.06.2019 19:00
How many moles of cu are needed to react with 5.8 moles of agno3? cu + 2 agno3 → cu(no3)2 + 2 ag
Answers: 3
You know the right answer?
Aqueous copper(II) nitrate reacts with aqueous sodium sulfide to produce aqueous sodium nitrate and...
Questions


Arts, 20.10.2019 20:10

Health, 20.10.2019 20:10



Chemistry, 20.10.2019 20:10


Social Studies, 20.10.2019 20:10



Social Studies, 20.10.2019 20:10

Mathematics, 20.10.2019 20:10


Biology, 20.10.2019 20:10

History, 20.10.2019 20:10

Mathematics, 20.10.2019 20:20


Mathematics, 20.10.2019 20:20