Iron(ii) sulfide reacts with hydrochloric acid according to the reaction:
fes(s)+2hcl(aq)→fe...

Chemistry, 17.10.2019 03:30 amberchule9681
Iron(ii) sulfide reacts with hydrochloric acid according to the reaction:
fes(s)+2hcl(aq)→fecl2(s)+h2s(g)
a reaction mixture initially contains 0.240 mol fes and 0.646 mol hcl.
what amount (in moles) of the excess reactant is left?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which of the following elements is a representative element? a. chromium (cr) b. aluminum (al) c. mercury (hg) d. silver (ag)
Answers: 1

Chemistry, 22.06.2019 22:30
Write and balance the chemical equation that represents the reaction of aqueous sulfuric acid with aqueous sodium hydroxide to form water and sodium sulfate. include phases.
Answers: 1

Chemistry, 22.06.2019 22:30
Which of these statements best explains why space exploration should be encouraged? it prepares humans to live without oxygen. it dispel myths about objects in space. it prevents comets and asteroids from striking earth. it creates technology to absorb harmful radiations in space.
Answers: 1

Chemistry, 23.06.2019 03:00
Describe the properties of sodium, chlorine, and sodium chloride
Answers: 1
You know the right answer?
Questions





Mathematics, 09.12.2020 22:40




Mathematics, 09.12.2020 22:40

Mathematics, 09.12.2020 22:40

Business, 09.12.2020 22:40

Mathematics, 09.12.2020 22:40






Mathematics, 09.12.2020 22:40


Mathematics, 09.12.2020 22:40

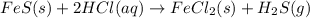
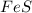 react with 2 mole of
react with 2 mole of 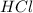
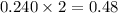 moles of
moles of 

