
Chemistry, 19.11.2020 05:40 SmartKitty
The graph represents the reaction 2H2 +02 = 2H20 as it reaches equilibrium. Based on the graph, which two statements about this reaction are true?
A. At point 2, the concentrations of H20, 02, and H2 are all changing toward their equilibrium concentrations.
B. The rate of formation of products is equal to the rate of formation of reactants only after point 4.
C. After point 2, the rate of formation of products is equal to the rate of formation of reactants.
D. At point 1, more reactants are converted to products than products are converted to reactants.
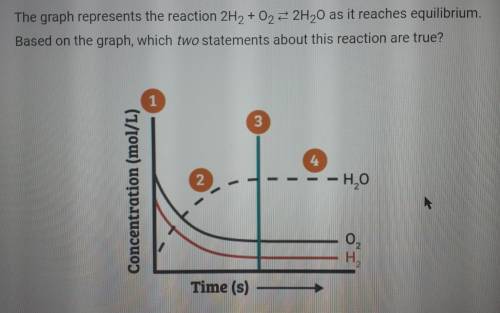

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 11:40
Which type of precipitation would most likely form when the surface air temperature is slightly below freezing and the air temperature increases as you move upward away from the ground?
Answers: 2

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 22.06.2019 14:30
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1
You know the right answer?
The graph represents the reaction 2H2 +02 = 2H20 as it reaches equilibrium. Based on the graph, whic...
Questions


Mathematics, 10.04.2021 07:50


Mathematics, 10.04.2021 07:50



Social Studies, 10.04.2021 07:50

Business, 10.04.2021 07:50







English, 10.04.2021 07:50

Biology, 10.04.2021 07:50

Mathematics, 10.04.2021 07:50



Physics, 10.04.2021 07:50



