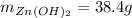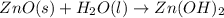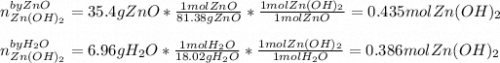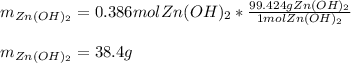Chemistry, 18.11.2020 17:10 kbkbkbkb7611
For the following reaction, 35.4 grams of zinc oxide are allowed to react with 6.96 grams of water . zinc oxide(s) + water(l) zinc hydroxide(aq) What is the maximum mass of zinc hydroxide that can be formed?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2

Chemistry, 22.06.2019 23:00
What is the average rate of the reaction between 10 and 20 s?
Answers: 1
You know the right answer?
For the following reaction, 35.4 grams of zinc oxide are allowed to react with 6.96 grams of water ....
Questions

Computers and Technology, 19.02.2020 04:56


English, 19.02.2020 04:56

Mathematics, 19.02.2020 04:56










Mathematics, 19.02.2020 04:56


Social Studies, 19.02.2020 04:57