
Copper metal (Cu) reacts with silver nitrate (AgNO3) in aqueous solution to form Ag and Cu(NO3)2. An excess of AgNO3 is present. The balanced chemical equation is shown below.
Cu + 2AgNO3 Right arrow. Cu(NO3)2 + 2Ag
The molar mass of Cu is 63.5 g/mol. The molar mass of Ag is 107.9 g/mol. What mass, in grams, of Ag is produced from reaction of 31.75 g of Cu?
26.95
107.9
215.91
431.82

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 16:30
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2

Chemistry, 23.06.2019 04:30
Two liquids are poured into a beaker. after a few seconds, the beaker becomes warm. which of the following best describes this reaction? a. an exothermic reaction b. a decomposition reaction c. an endothermic reaction d. a single-displacement reaction
Answers: 1
You know the right answer?
Copper metal (Cu) reacts with silver nitrate (AgNO3) in aqueous solution to form Ag and Cu(NO3)2. An...
Questions

Geography, 28.10.2020 14:00

Spanish, 28.10.2020 14:00

Chemistry, 28.10.2020 14:00

Arts, 28.10.2020 14:00


Social Studies, 28.10.2020 14:00

English, 28.10.2020 14:00

Mathematics, 28.10.2020 14:00



English, 28.10.2020 14:00

Mathematics, 28.10.2020 14:00


Social Studies, 28.10.2020 14:00



Arts, 28.10.2020 14:00

History, 28.10.2020 14:00

English, 28.10.2020 14:00

Mathematics, 28.10.2020 14:00

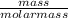
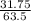 = 0.5moles
= 0.5moles 

