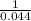19. The reaction 2NOBr (g) → 2 NO (g) + Br2 (g) is a second-order reaction with a rate constant of 0.80 M-1s-1 at 11 °C. If the initial concentration of NOBr is 0.0440 M, the concentration of NOBr after 6.0 seconds is . A) 0.0276 M B) 0.0324 M C) 0.0363 M D) 0.0348 M E) 0.0402 M

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1
You know the right answer?
19. The reaction 2NOBr (g) → 2 NO (g) + Br2 (g) is a second-order reaction with a rate constant of 0...
Questions


Mathematics, 20.02.2021 01:00

Health, 20.02.2021 01:00


Mathematics, 20.02.2021 01:00

Mathematics, 20.02.2021 01:00


Mathematics, 20.02.2021 01:00





Chemistry, 20.02.2021 01:00


Mathematics, 20.02.2021 01:00




History, 20.02.2021 01:00

Physics, 20.02.2021 01:00

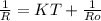
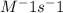
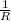 = 0.8*6+
= 0.8*6+