
Chemistry, 17.11.2020 01:00 JvGaming2001
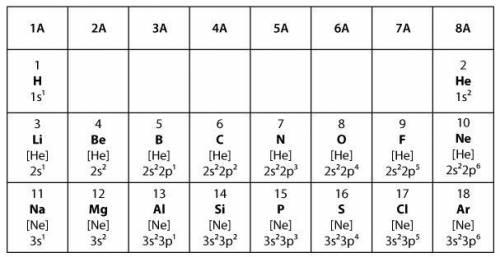
Covalent bonds form when two elements share electrons to make a complete outer shell of electrons. Ionic bonds form when one element donates one or more electrons to another element to make a complete outer shell of electrons.
Which statement best explains the type of bond that will form between two elements from group 6A in the model?
A
The two elements will form a covalent bond because both elements will share a single electron in order to have full outer shells.
B
The two elements will form a covalent bond because both elements will share a pair of electrons in order to have full outer shells.
C
The two elements will form an ionic bond because one of the elements will donate one electron to the other element in order to have full outer shells.
D
The two elements will form an ionic bond because one of the elements will donate two electrons to the other element in order to have full outer shells.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2


Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
Covalent bonds form when two elements share electrons to make a complete outer shell of electrons. I...
Questions



Health, 01.07.2021 18:10









Business, 01.07.2021 18:10

Computers and Technology, 01.07.2021 18:10





Mathematics, 01.07.2021 18:10




