

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have metallic bonds. which of the following is most likely a property of this substance? a. low conductivity b. low boiling point c. high malleability d. high solubility in water
Answers: 2

Chemistry, 23.06.2019 04:31
Chemical engineering who specializes in negotiating for large purchases and instructing customers in use of the products are
Answers: 1

Chemistry, 23.06.2019 09:30
Need , hurry pls create a superhero out of the element iron, what are its powers and his sidekick ( an element that works well with iron). how was the superhero made and who discovered him
Answers: 3
You know the right answer?
One of the light waves produced when hydrogen is energized has a wavelength of 4.105 x10^-7 m. What...
Questions

Mathematics, 04.12.2020 20:20

Mathematics, 04.12.2020 20:20


Mathematics, 04.12.2020 20:20

Mathematics, 04.12.2020 20:20


Mathematics, 04.12.2020 20:20

History, 04.12.2020 20:20



Mathematics, 04.12.2020 20:20

Physics, 04.12.2020 20:20



Social Studies, 04.12.2020 20:20



Mathematics, 04.12.2020 20:20

Physics, 04.12.2020 20:20

English, 04.12.2020 20:20

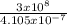 = 7.3 x 10¹⁴Hz
= 7.3 x 10¹⁴Hz

