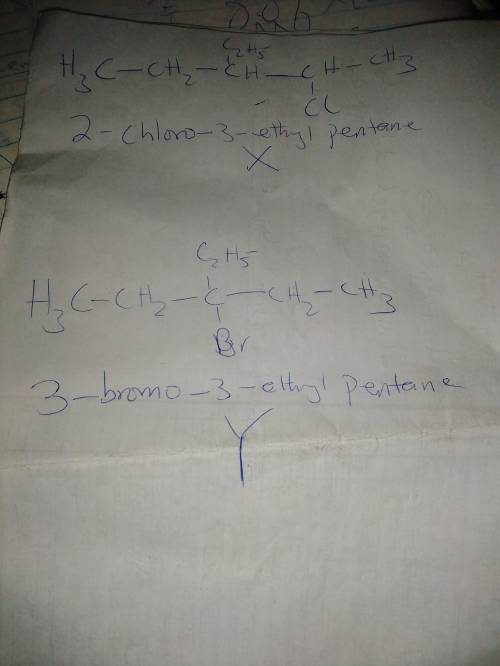Chemistry, 16.11.2020 16:50 crispingolfer1864
Compounds X has the formula C7H15Cl; Y is C7H15Br. X undergoes base-promoted E2 elimination to give a single alkene product Z. Y likewise reacts under similar conditions to give a single alkene product that is isomeric with Z Catalytic hydrogenation of Z affords 3-ethylpentane. X readily reacts in SN2 fashion with sodium iodide in acetone. Y does not undergo a similar SN2 reaction. Propose structures for X and Y.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 04:00
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 23.06.2019 01:00
If a sample of radioactive isotopes takes 600 minutes to decay from 400 grams to 50 grams, what is the half-life of the isotope?
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Compounds X has the formula C7H15Cl; Y is C7H15Br. X undergoes base-promoted E2 elimination to give...
Questions

Mathematics, 16.10.2020 16:01



English, 16.10.2020 16:01


English, 16.10.2020 16:01


Mathematics, 16.10.2020 16:01

Mathematics, 16.10.2020 16:01




Mathematics, 16.10.2020 16:01



History, 16.10.2020 16:01



Mathematics, 16.10.2020 16:01