
Chemistry, 13.11.2020 23:20 vanessa791
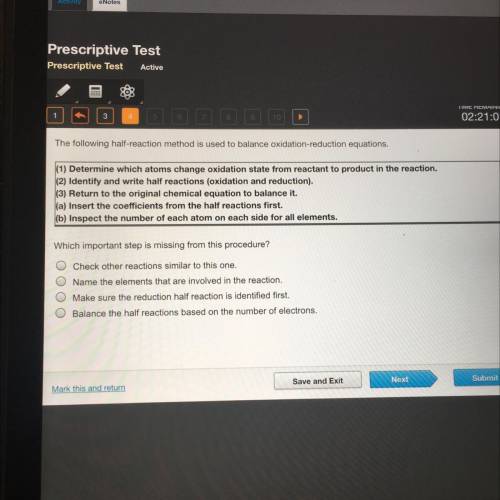
The following half-reaction method is used to balance oxidation-reduction equations.
(1) Determine which atoms change oxidation state from reactant to product in the reaction.
(2) Identify and write half reactions (oxidation and reduction).
(3) Return to the original chemical equation to balance it.
(a) Insert the coefficients from the half reactions first.
(b) Inspect the number of each atom on each side for all elements.
Which important step is missing from this procedure?
Check other reactions similar to this one.
Name the elements that are involved in the reaction.
Make sure the reduction half reaction is identified first.
Balance the half reactions based on the number of electrons.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Choose all the answers that apply. ionic compounds dissolve easily in water do not dissolve in water have low melting points have high melting points conduct electricity when melted
Answers: 1

Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1


Chemistry, 23.06.2019 00:30
Which of the following best describes technology a. something created for only scientists to use b.the method of thinking that scientists use. c.the application of engineering to create useful products. c. a scientific idea
Answers: 1
You know the right answer?
The following half-reaction method is used to balance oxidation-reduction equations.
(1) Determine...
Questions




Mathematics, 27.07.2021 02:00



Mathematics, 27.07.2021 02:00

Mathematics, 27.07.2021 02:00















