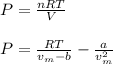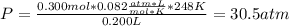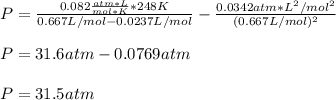Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 12:00
Marcel just purchased 1.69 grams of iron fillings in order to make living putty for his 6 year old niece. how many moles of iron are made in his sample?
Answers: 1

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1
You know the right answer?
For a 0.300 mol sample of helium gas in a 0.200 L container at 248K, will the pressure be greater if...
Questions

Biology, 26.02.2021 03:00


English, 26.02.2021 03:00


Biology, 26.02.2021 03:00

Computers and Technology, 26.02.2021 03:00

History, 26.02.2021 03:00


History, 26.02.2021 03:00

Mathematics, 26.02.2021 03:00

History, 26.02.2021 03:00


Mathematics, 26.02.2021 03:00




Mathematics, 26.02.2021 03:00

Biology, 26.02.2021 03:00

English, 26.02.2021 03:00