
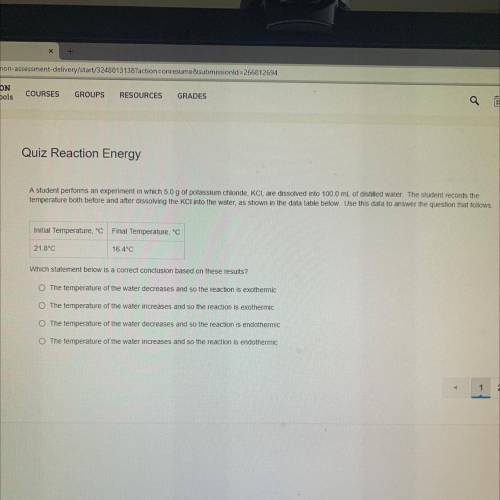
A student performs an experiment in which 5.0 g of potassium chloride, KCI, are dissolved into 100.0 mL of distilled water. The student records the
temperature both before and after dissolving the KCl into the water, as shown in the data table below. Use this data to answer the question that follows.
Initial Temperature, °C Final Temperature, °C
21.8°C
16.4°C
Which statement below is a correct conclusion based on these results ?
The temperature of the water decreases and so the reaction is exothermic
The temperature of the water increases and so the reaction is exothermic
The temperature of the water decreases and so the reaction is endothermic
The temperature of the water increases and so the reaction is endothermic

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 23.06.2019 01:30
The solubility of barium nitrate is 9.02 g/100 g h2o at 20°c. a 15.2 g sample of barium nitrate is added to 200.0 g of water at 20°c. is the solution saturated, unsaturated, or supersaturated? a. unsaturated b. saturated c. supersaturated
Answers: 1
You know the right answer?
A student performs an experiment in which 5.0 g of potassium chloride, KCI, are dissolved into 100.0...
Questions

Mathematics, 23.10.2020 20:10

Mathematics, 23.10.2020 20:10

Mathematics, 23.10.2020 20:10

Mathematics, 23.10.2020 20:10





Biology, 23.10.2020 20:10

Geography, 23.10.2020 20:10

Engineering, 23.10.2020 20:10


Mathematics, 23.10.2020 20:10

Mathematics, 23.10.2020 20:10

Mathematics, 23.10.2020 20:10



Mathematics, 23.10.2020 20:10

Mathematics, 23.10.2020 20:10



