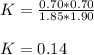Chemistry, 11.11.2020 17:50 karlacarreras152
For the following reaction at equilibrium SO3(g) + NO(g) = SO2(g) + NO2(g)It is found that [SO2] = 0.70 M and [NO] = 1.20 M. Calculate the equilibrium constant for the readction knowing that the initial concentration were [SO3] = 2.55 M and [NO] = 1.90 M.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2

Chemistry, 22.06.2019 08:00
Will give ! what are the advantages and disadvantages of nuclear power? check all that apply. one advantage of nuclear energy is that it does not produce carbon dioxide emissions. storage of nuclear waste is a short-term problem associated with nuclear energy. the problem with uranium mining is that a large quantity of uranium must be extracted to meet energy needs because the energy release from uranium fission is so low. safe operation of a nuclear power plant can be jeopardized by a human mistake.
Answers: 1

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3

Chemistry, 22.06.2019 16:00
As changes in energy levels of electrons increase, the frequencies of atomic line spectra they emit
Answers: 2
You know the right answer?
For the following reaction at equilibrium SO3(g) + NO(g) = SO2(g) + NO2(g)It is found that [SO2] = 0...
Questions

Mathematics, 28.03.2021 01:00



Mathematics, 28.03.2021 01:00

Mathematics, 28.03.2021 01:00


Mathematics, 28.03.2021 01:00







Mathematics, 28.03.2021 01:00

Mathematics, 28.03.2021 01:00

Chemistry, 28.03.2021 01:00




Mathematics, 28.03.2021 01:00

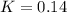
![K=\frac{[SO_2][NO_2]}{[SO_3][NO]}](/tpl/images/0887/9424/17768.png)
 is:
is: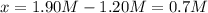
![K=\frac{x*x}{([SO_3]_0-x)([NO]_0-x)}](/tpl/images/0887/9424/71b6f.png)
![[SO_3]=2.55M-0.70M=1.85M](/tpl/images/0887/9424/debed.png)
![[NO]=1.20M](/tpl/images/0887/9424/e6bfc.png)
![[SO_2]=0.70M](/tpl/images/0887/9424/ae391.png)
![[NO_2]=0.70M](/tpl/images/0887/9424/98697.png)