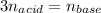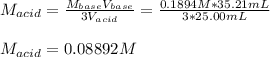Chemistry, 11.11.2020 17:50 montgomerykarloxc24x
Calculate the concentration (M) of arsenic acid (H3AsO4) in a solution if 25.00 mL of that solution required 35.21 mL of 0.1894 M KOH for neutralization.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1

Chemistry, 22.06.2019 22:00
4.25g sample of solid ammonium nitrate dissolves in 60.0g of water in a coffee-cup calorimeter, the temperature drops from 22.0 c to 16.9 c. assume that the specific heat of the solution is the same as that of pure water. calculate delta(h) (in kj/mol nh4no3) for the solution proces.
Answers: 2

You know the right answer?
Calculate the concentration (M) of arsenic acid (H3AsO4) in a solution if 25.00 mL of that solution...
Questions




Mathematics, 24.05.2020 17:58

Biology, 24.05.2020 17:58

Engineering, 24.05.2020 17:58

Physics, 24.05.2020 17:58




Mathematics, 24.05.2020 17:58






Computers and Technology, 24.05.2020 17:58

Law, 24.05.2020 17:58

Mathematics, 24.05.2020 17:58