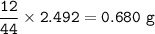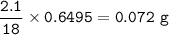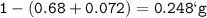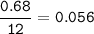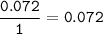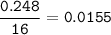Chemistry, 11.11.2020 16:00 HalpMehPlz
A 100g of sample of a compound is combusted in excess oxygen and the products are 2.492g of CO2 and 0.6495 of H2O. Determine the empirical formula of the compound?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3

Chemistry, 22.06.2019 17:30
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2
You know the right answer?
A 100g of sample of a compound is combusted in excess oxygen and the products are 2.492g of CO2 and...
Questions

Business, 23.03.2021 21:50

English, 23.03.2021 21:50




Chemistry, 23.03.2021 21:50

Biology, 23.03.2021 21:50


History, 23.03.2021 21:50

Mathematics, 23.03.2021 21:50


English, 23.03.2021 21:50


Social Studies, 23.03.2021 21:50



English, 23.03.2021 21:50

English, 23.03.2021 21:50


Mathematics, 23.03.2021 21:50