
2 The substance has a mass of 0.50 kg.
34 000 J of energy is transferred to the substance to completely melt it.
The temperature of the substance does not change during this time.
Calculate the specific latent heat of fusion of the substance.
Use an equation from the Equations List.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 14:00
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1


Chemistry, 22.06.2019 19:40
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests.which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3
You know the right answer?
2 The substance has a mass of 0.50 kg.
34 000 J of energy is transferred to the substance to comple...
Questions

Biology, 16.10.2020 09:01


History, 16.10.2020 09:01

History, 16.10.2020 09:01


Mathematics, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01


History, 16.10.2020 09:01



Mathematics, 16.10.2020 09:01

History, 16.10.2020 09:01

Arts, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01


History, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01

Arts, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01

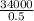 = 68000J/kg
= 68000J/kg

