
Chemistry, 11.11.2020 05:50 Dogtes9667
Calculate the pH of a buffer solution when 32.0 mL of a 0.25 M H2CO3 is mixed with 20.0 mL of a 0.15 M NaHCO3

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:20
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2(g)→2alcl3(s) what is the maximum mass of aluminum chloride that can be formed when reacting 32.0 g of aluminum with 37.0 g of chlorine? express your answer to three significant figures and include the appropriate units.
Answers: 2

Chemistry, 21.06.2019 17:00
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 21.06.2019 18:40
Determine the mass of fuel required for the expected energy consumption in the united states for the next ten years. energy use per person per year in the united states = 3.5 x 1011joules base calculations on current population of 310,000,000.
Answers: 2

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3
You know the right answer?
Calculate the pH of a buffer solution when 32.0 mL of a 0.25 M H2CO3 is mixed with 20.0 mL of a 0.15...
Questions



Mathematics, 20.02.2021 02:30


Mathematics, 20.02.2021 02:30


Mathematics, 20.02.2021 02:30

Mathematics, 20.02.2021 02:30



Mathematics, 20.02.2021 02:30



History, 20.02.2021 02:30


Mathematics, 20.02.2021 02:30



Mathematics, 20.02.2021 02:30

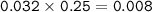
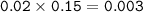
![\tt [H^+]=Ka\dfrac{mole~weak~acid}{mole~salt}\\\\(H^+]=1.8\times 10^{-5}\dfrac{0.008}{0.003}=4.8\times 10^{-5}\\\\pH=5-log~4.8=4.32](/tpl/images/0887/1834/e5293.png)


