Chemistry, 10.11.2020 20:10 aydenmasonc
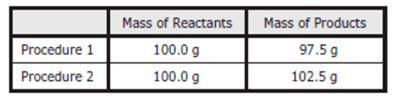
As part of an investigation, students combined substances in a beaker to observe chemical reactions. They performed two procedures. They measured the mass of each substance before and after each reaction. The table shows their observations. Assuming the students did not make any careless errors, what likely explains these changes in mass?
Question 3 options:
Procedure 1: All the reactants were liquids that evaporated. Procedure 2: A gas was formed as one product, and it escaped into the air.
Procedure 1: One of the reactants was converted to thermal energy. Procedure 2: All the products were liquids.
Procedure 1: The reactants were liquids with different densities. Procedure 2: The reactants were combined into only one product.
Procedure 1: One of the products was a gas that escaped into the air. Procedure 2: A gas from the air reacted with one of the other reactants.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2

Chemistry, 22.06.2019 21:30
What is the correct name for the compound cocl3? a) cobalt(i) chloride b) cobalt(i) chlorate c) cobalt(ii) chlorate d) cobalt(iii) chloride
Answers: 1

Chemistry, 23.06.2019 06:40
8. how much enthalpy/heat is transferred when 0.5113gof ammonia (nh3) reacts with excess oxygen according| to the following equation: 4nh3 +502 - 4n0+ 6h20ah = -905.4j
Answers: 1
You know the right answer?
As part of an investigation, students combined substances in a beaker to observe chemical reactions....
Questions

Biology, 17.01.2020 04:31

Mathematics, 17.01.2020 04:31



Computers and Technology, 17.01.2020 04:31






Computers and Technology, 17.01.2020 04:31

Social Studies, 17.01.2020 04:31