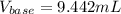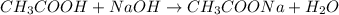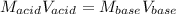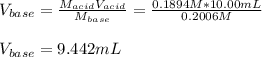Chemistry, 09.11.2020 16:50 brooke7768
Given that 10.00mL of 0.1894M CH3COOH was titrated with 0.2006M NaOH in this experiment, calculate the volume, in mL, of NaOH required

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 13:00
In a copper wire, a temperature increase is the result of which of the following
Answers: 1

Chemistry, 22.06.2019 13:30
What produces wave a)sound b) heats c)transfer of energy d)vibrations
Answers: 2
You know the right answer?
Given that 10.00mL of 0.1894M CH3COOH was titrated with 0.2006M NaOH in this experiment, calculate t...
Questions

English, 13.08.2019 12:10





Health, 13.08.2019 12:10


Mathematics, 13.08.2019 12:10


Biology, 13.08.2019 12:10

Business, 13.08.2019 12:10

Mathematics, 13.08.2019 13:10



Mathematics, 13.08.2019 14:10


Biology, 13.08.2019 14:10

Mathematics, 13.08.2019 14:10

Mathematics, 13.08.2019 14:10

Health, 13.08.2019 14:10