
Chemistry, 07.11.2020 15:40 delgadillogiselle711
If a sample containing 18.1 g of NH3 is reacted with 90.4 g of
Cuo, which is the limiting reactant? How many grams of N2
will be formed?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 23.06.2019 04:00
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 3

Chemistry, 23.06.2019 10:00
An uncovered pot of water lies out in the sun. which statements correctly describe what happens at the surface of the liquid water? 1. the vapor pressure remains constant regardless of the water temperature. 2. the vapor pressure is produced by water molecules that have evaporated. 3. the vapor pressure increases as the sun heats the water in the pot. 4. evaporation stops once the vapor pressure reaches a certain point. 5. evaporation and condensation both occur on the liquid’s surface.
Answers: 3
You know the right answer?
If a sample containing 18.1 g of NH3 is reacted with 90.4 g of
Cuo, which is the limiting reactant?...
Questions

Social Studies, 13.10.2019 11:30


Mathematics, 13.10.2019 11:30




Mathematics, 13.10.2019 11:30


History, 13.10.2019 11:30

Mathematics, 13.10.2019 11:30

Health, 13.10.2019 11:30




Biology, 13.10.2019 11:30


World Languages, 13.10.2019 11:30


Mathematics, 13.10.2019 11:30

English, 13.10.2019 11:30

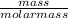
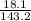 = 0.13moles
= 0.13moles 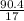 = 5.32moles
= 5.32moles 

