1 point
Why DOESN'T the equation pictured satisfy the Law of Conservation of
Matter?
H2...

Chemistry, 07.11.2020 07:00 edgartorres5123
1 point
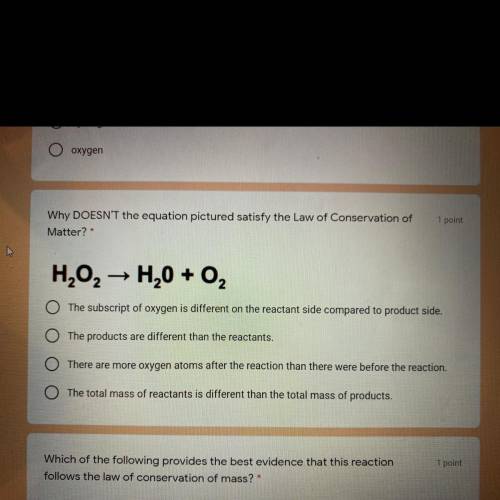
Why DOESN'T the equation pictured satisfy the Law of Conservation of
Matter?
H202 → H20 + O2
The subscript of oxygen is different on the reactant side compared to product side.
The products are different than the reactants.
There are more oxygen atoms after the reaction than there were before the reaction.
The total mass of reactants is different than the total mass of products.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 22:30
How do limiting factors most affect population size? ostop population growthrestrict population growthincrease population sizeresult in positive impactso
Answers: 1

Chemistry, 23.06.2019 01:30
Polar bears give birth and hunt on sea ice. which of the following would polar bears survive during the melting of arctic ice? growing another layer of fur during summer migrate inland to search for different food sources staying put until the ice refreezes sticking to the usual diet of seals
Answers: 1

Chemistry, 23.06.2019 11:30
Which of these have the same number of particles as 1 mole of water h2o
Answers: 1
You know the right answer?
Questions



Chemistry, 27.06.2019 11:00

Spanish, 27.06.2019 11:00


World Languages, 27.06.2019 11:00






History, 27.06.2019 11:00


English, 27.06.2019 11:00




Health, 27.06.2019 11:00

Mathematics, 27.06.2019 11:00



