
How would an element on the left side of row 2 of the periodic table differ from an element in the middle of the same row?
A. The element on the left would have more atomic mass.
B. The element on the left would have less malleability.
C. The element on the left would have a lower melting point.
D. The element on the left would have no metallic properties.
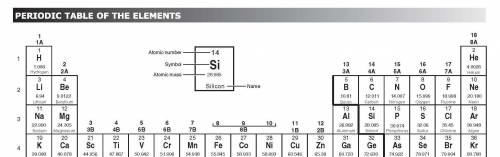

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1


Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2
You know the right answer?
How would an element on the left side of row 2 of the periodic table differ from an element in the m...
Questions


Mathematics, 02.05.2021 23:50

Chemistry, 02.05.2021 23:50

History, 02.05.2021 23:50

Mathematics, 02.05.2021 23:50




Biology, 02.05.2021 23:50

English, 02.05.2021 23:50


Business, 02.05.2021 23:50



Mathematics, 02.05.2021 23:50

Biology, 02.05.2021 23:50


English, 02.05.2021 23:50


Computers and Technology, 02.05.2021 23:50



