
Chemistry, 06.11.2020 20:00 thibeauxkristy
A sample of 87.6 g of carbon is reacted with 136 g of
fluorine gas to produce carbon tetrafluoride. Using
the balanced equation below, predict which is the
limiting reactant and the maximum amount in moles
of carbon tetrafluoride that can be produced.
C +2F2 → CF4
A. fluorine, 1.79 moles
B. carbon, 7.29 moles
C. fluorine, 6.72 moles
D. carbon, 4.63 moles

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3
You know the right answer?
A sample of 87.6 g of carbon is reacted with 136 g of
fluorine gas to produce carbon tetrafluoride....
Questions





English, 05.05.2020 18:01


Biology, 05.05.2020 18:01



Mathematics, 05.05.2020 18:01

Biology, 05.05.2020 18:01

English, 05.05.2020 18:01

Mathematics, 05.05.2020 18:01

Computers and Technology, 05.05.2020 18:01

English, 05.05.2020 18:01





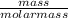
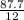 = 7.31moles
= 7.31moles 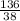 = 3.58moles
= 3.58moles 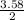 = 1.79moles of CF₄
= 1.79moles of CF₄

