
Chemistry, 06.11.2020 17:10 delanieloya
Suppose that 0.410 mol of methane, CH4(g), is reacted with 0.560 mol of fluorine, F2(g), forming CF4(g) and HF(g) as sole products. Assuming that the reaction occurs at constant pressure, how much heat is released

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:40
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1

Chemistry, 22.06.2019 17:30
In a heat of an engine, if 700 j enters the system, and the piston does 400 j of work what is the final internal (thermal) energy of the system if the initial energy is 1,500 j
Answers: 2

Chemistry, 22.06.2019 19:00
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
You know the right answer?
Suppose that 0.410 mol of methane, CH4(g), is reacted with 0.560 mol of fluorine, F2(g), forming CF4...
Questions


Mathematics, 10.07.2019 17:20




Mathematics, 10.07.2019 17:20






English, 10.07.2019 17:20




English, 10.07.2019 17:20




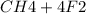 →
→ 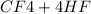
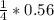 = 0.14mole of CF44 moles of fluorine → 4 moles of HF0.56 mole of fluorine → 0.56 mole of HF
= 0.14mole of CF44 moles of fluorine → 4 moles of HF0.56 mole of fluorine → 0.56 mole of HF

