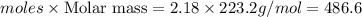Chemistry, 06.11.2020 16:40 ahnaodoido384
An excess of oxygen reacts with 451.4 g of lead, forming 374.7 g of lead(II) oxide. Calculate the percent yield of the reaction.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1

Chemistry, 23.06.2019 05:30
Based on the formulas, select the compounds below that are covalent: kbr sif4 al2o3 co2 naco3 s7o2 pcl3 fe3n2 h2o s2f10
Answers: 3
You know the right answer?
An excess of oxygen reacts with 451.4 g of lead, forming 374.7 g of lead(II) oxide. Calculate the pe...
Questions



Mathematics, 05.06.2020 21:05



Mathematics, 05.06.2020 21:05


Mathematics, 05.06.2020 21:05

Mathematics, 05.06.2020 21:05

English, 05.06.2020 21:05

Mathematics, 05.06.2020 21:05






History, 05.06.2020 21:05



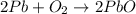
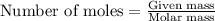
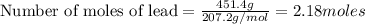
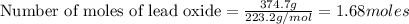
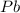 produces = 2 moles of
produces = 2 moles of 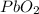
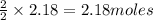 of
of