Lab Reaction Rate :
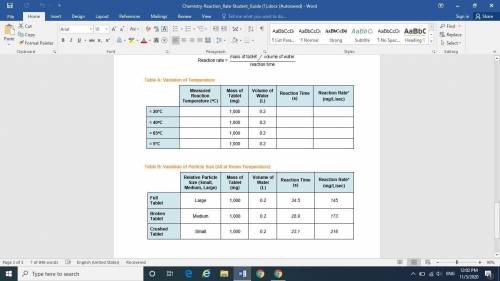
Variation of Temperature
Step 2: Measure the Reaction Rate at ≈ 20°C (Roo...

Chemistry, 05.11.2020 20:30 CloutLEVEL
Lab Reaction Rate :
Variation of Temperature
Step 2: Measure the Reaction Rate at ≈ 20°C (Room Temperature)
Step 3: Measure the Reaction Rate at ≈ 40°C
Step 4: Repeat Step 2, heating the water to Measure the Reaction Rate at ≈ 65°C
Step 5: Measure the Reaction Rate at ≈ 5°C
Variation of Particle Size
Step 6: Measure the Reaction Rate for a Full Tablet
Step 7: Measure the Reaction Rate for a Partially Broken Tablet
Step 8: Measure the Reaction Rate for a Crushed Tablet


Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:30
The chart shows the bid provided by four contractors to complete a job. which contractor is the most cost-effective?
Answers: 3

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 22:30
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1

Chemistry, 23.06.2019 05:00
How many atomic mass units are equal to 1.672×10−24 g of protons?
Answers: 3
You know the right answer?
Questions









Mathematics, 04.05.2021 16:50




Law, 04.05.2021 16:50

Mathematics, 04.05.2021 16:50

Mathematics, 04.05.2021 16:50



Mathematics, 04.05.2021 16:50




