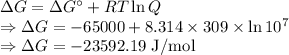Calculate the standard biological Gibbs free energy for the reaction: pyruvate- + NADH + H+(aq) ---> Lactate- + NAD+ at 309 K given that the standard Gibbs free energy = -65.0 kJ/mol at this temperature. This reaction occurs under conditions of low oxygen supply, such as in muscle cells during strenuous exercise. Note: See Box 7.1 on page 164. The biological standard state has hydrogen ions at 1x10-7 molar instead of 1 M.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:30
1. baking powder is a 1: 1 molar mixture of cream of tartar (khc4h4o6) and baking soda (nahco3). a recipe calls for two teaspoons (a total of 8.0 grams) of cream of tartar. how much baking soda must be added for both materials to react completely?
Answers: 2

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1

Chemistry, 22.06.2019 12:30
Consider the four elements above. which one of these elements will combine with oxygen in a 1: 1 ratio?
Answers: 3
You know the right answer?
Calculate the standard biological Gibbs free energy for the reaction: pyruvate- + NADH + H+(aq) ---&...
Questions

Mathematics, 04.12.2021 09:30

Engineering, 04.12.2021 09:30

English, 04.12.2021 09:30

Geography, 04.12.2021 09:30

Social Studies, 04.12.2021 09:30


Mathematics, 04.12.2021 09:40



History, 04.12.2021 09:40

Mathematics, 04.12.2021 09:40



World Languages, 04.12.2021 09:40


Mathematics, 04.12.2021 09:40




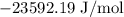
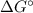 = Standard Gibbs free energy = -65.0 kJ/mol
= Standard Gibbs free energy = -65.0 kJ/mol![[H^+]](/tpl/images/0870/7831/07acb.png) = Biological standard state has hydrogen ions =
= Biological standard state has hydrogen ions = 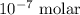
![Q=\dfrac{1}{[H^+]}\\\Rightarrow Q=\dfrac{1}{10^{-7}}\\\Rightarrow Q=10^7](/tpl/images/0870/7831/5fb93.png)