
Chemistry, 04.11.2020 19:00 JaylenGuidish
(see attachment)
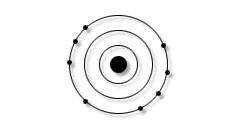
This element has 3 energy shells. The outer shell of this element was determined as illustrated here.
According to the Periodic Table of Elements, it must be:
Beryllium (Be)
Nitrogen (N)
Argon (Ar)
Oxygen (O)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2


Chemistry, 23.06.2019 06:20
Examine the false statement. compounds are the smallest unit of an element that occur most commonly in nature. select the rewording of the statement that is true. a: atoms are the smallest unit of an element that commonly occur in nature. b: molecules are the smallest unit of an element or compound that commonly occur in nature. c: molecules are the smallest unit of a compound that occur on the periodic table. d: compounds are the smallest unit of an element that occur on the periodic table
Answers: 1
You know the right answer?
(see attachment)
This element has 3 energy shells. The outer shell of this element was determined a...
Questions

Mathematics, 14.12.2021 03:10



Mathematics, 14.12.2021 03:10


English, 14.12.2021 03:10

History, 14.12.2021 03:10

SAT, 14.12.2021 03:10

Chemistry, 14.12.2021 03:10




English, 14.12.2021 03:10


Mathematics, 14.12.2021 03:10

Advanced Placement (AP), 14.12.2021 03:10

Mathematics, 14.12.2021 03:10





