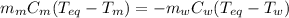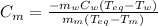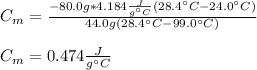Chemistry, 03.11.2020 17:20 Justadumbemo
A 44.0 g sample of an unknown metal at 99.0 oC was placed in a constant-pressure calorimeter of negligible heat capacity containing 80.0 mL water at 24.0 oC. The final temperature of the system was found to be 28.4 oC. Calculate the Specific heat of the metal if density of water is 1.00 g/ml.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 23:00
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2

Chemistry, 23.06.2019 03:00
The size (radius) of an oxygen molecule is about 2.0 ×10−10m. make a rough estimate of the pressure at which the finite volume of the molecules should cause noticeable deviations from ideal-gas behavior at ordinary temperatures (t= 300k ). assume that deviatons would be noticeable when volume of the gas per molecule equals the volume of the molecule itself.
Answers: 3
You know the right answer?
A 44.0 g sample of an unknown metal at 99.0 oC was placed in a constant-pressure calorimeter of negl...
Questions



Chemistry, 06.10.2019 10:02

Biology, 06.10.2019 10:02

Mathematics, 06.10.2019 10:02

Social Studies, 06.10.2019 10:02

Chemistry, 06.10.2019 10:02

Mathematics, 06.10.2019 10:02


Physics, 06.10.2019 10:02



Social Studies, 06.10.2019 10:02




Chemistry, 06.10.2019 10:02

Geography, 06.10.2019 10:02